Our Projects

Myocure Project
The objective of this project is to find therapeutic options for congenital myopathies.
Congenital myopathies are a group of genetic muscle diseases that are classified based on the histopathological characteristics observed in muscle biopsy.
Nemaline myopathy (NM), which is part of this group of diseases, was described for the first time as a non-progressive congenital musculoskeletal disorder, characterized by the presence of inclusions in muscle fibers called “nemalinic rods”. This pathology contains a wide genetic heterogeneity that produces a similar phenotype. Its incidence is 1 in 50,000 live births; although in our country the incidence of this pathology is unknown, since in clinical practice it is a rare disease. Mutations have been found in more than 10 different genes that cause the disease; seven of which code for sarcomeric thin filament components (NEB, ACTA1, TPM2, TPM3, TNNT1, CFL2, LMOD3) and 3 genes (KBTBD13, KLHL40 and KLHL41), belonging to the BTB-BACK-kelch family of proteins (BBK) involved in the ubiquitin-proteasome pathway.
The objectives of the Myocure platform are: (1) To establish cellular models to understand the pathophysiological mechanisms of NM. (2) To identify potential therapies by developing a pharmacological screening methodology in cell models of NM.
As a screening strategy, we assess the correct formation of actin filaments by fluorescence microscopy techniques. Positive compounds are those that are capable of restoring the correct formation of actin filaments in patient’s fibroblasts. Next, favorable compounds are confirmed by verifying the improvement of all pathophysiological alterations. A collection of compounds are routinely tested.
Currently, the Myocure platform is performing precision medicine in 8 patients with nemaline myopathy. We have recently obtained positive results in the pharmacological screening carried out on the fibroblasts of the patients. Therefore, we are currently working on the direct reprogramming of skin fibroblasts to skeletal muscle cells in order to evaluate the effect of positive compounds on the cell type that is mainly affected by the disease: the skeletal muscle cell.
The patient association “Yo Nemalínica” participates in the financing of this project. “Yo nemalínica” is a Spanish association of people affected by nemaline myopathy and their families, currently made up of 27 families, including a total of 28 affected people. This is the link to the association’s website, which also has an online store to contribute to fundraising.

Image of the visit of the president of the “Yo Nemalínica” Association (José Manuel Palomo) to our laboratory

Braincure Project
Initially, the Braincure project was developed for performing precision or personalized medicine in rare neurodegenerative diseases with brain iron accumulation. Nowadays, the Braincure project includes a large number of neurodegenerative diseases, such as Friedreich’s ataxia, Hereditary spastic ataxias, Amyotrophic lateral sclerosis and Huntington’s disease. However, since they are all neurodegenerative diseases, the precision medicine strategy is very similar for all of them.
The objective of this platform is to deepen the pathophysiology of the diseases and find effective personalized treatments using fibroblasts and neuronal cells derived from patients.
For this, we characterize the pathophysiological mechanisms and evaluate the effectiveness of a library of commercial pharmacological compounds in the recovery of pathological alterations in the patient-derived cells. In a first stage, pharmacological screening is carried out in fibroblasts derived from patients. For high throughput screening, a library of 426 drugs approved by the U.S. FDA (United States Food Drug Administration) from Selleck Chemicals (Houston, TX, USA) including a selection of compounds frequently used in patients treatments (iron chelators, antioxidants, pantothenate, creatine etc.) are routinely evaluated. To evaluate the effect of each testing compound, we carry out in deep evaluation of iron metabolism alterations, expression levels of mutant enzymes, mitochondrial dysfunction, oxidative stress, lipid metabolism and spontaneous and induced apoptosis in patients’s fibroblasts after treatment, comparing the obtained results with the results of patients’s fibroblasts without treatment and fibroblasts from healthy patients. In parallel, induced neurons (INs) are generated by direct reprogramming from patient fibroblasts. The most favorable compounds in fibroblasts screening are selected for testing in mutant neurons iNs.
Diseases included in the Braincure project:
Neurodegeneration with brain iron accumulation (NBIA)
Neurodegeneration with brain iron accumulation (NBIA) is a group of rare neurodegenerative disorders of genetic origin, characterized by a dysfunction of the central nervous system and the accumulation of iron in certain areas of the brain that causes the progressive disability of patients. Most NBIAs begin clinically in childhood and are inherited with an autosomal recessive pattern. Currently, there are no effective treatments for the great majority of these diseases.
Braincure platform performs precision medicine in the most prevalent NBIA disorders: PKAN, pantothenate kinase-associated neurodegeneration, caused by mutations in the PANK2 gene; PLAN, PLA2G6-associated neurodegeneration, due to mutations in the PLA2G6 gene; BPAN, Beta-propeller protein-associated neurodegeneration, caused by mutations in the WDR45 gene; MPAN, Mitochondrial-membrane Protein-associated Neurodegeneration, due to mutations in the C19orf12 gene; and FHAN, Fatty acid hydroxylase-associated neurodegeneration, caused by mutations in the FAH2 gene). We are currently performing personalized medicine in more than 50 patients from both Spain and abroad (Brazil, Colombia, Mexico, USA, France, United Kingdom, Holland, Hungary and Poland).
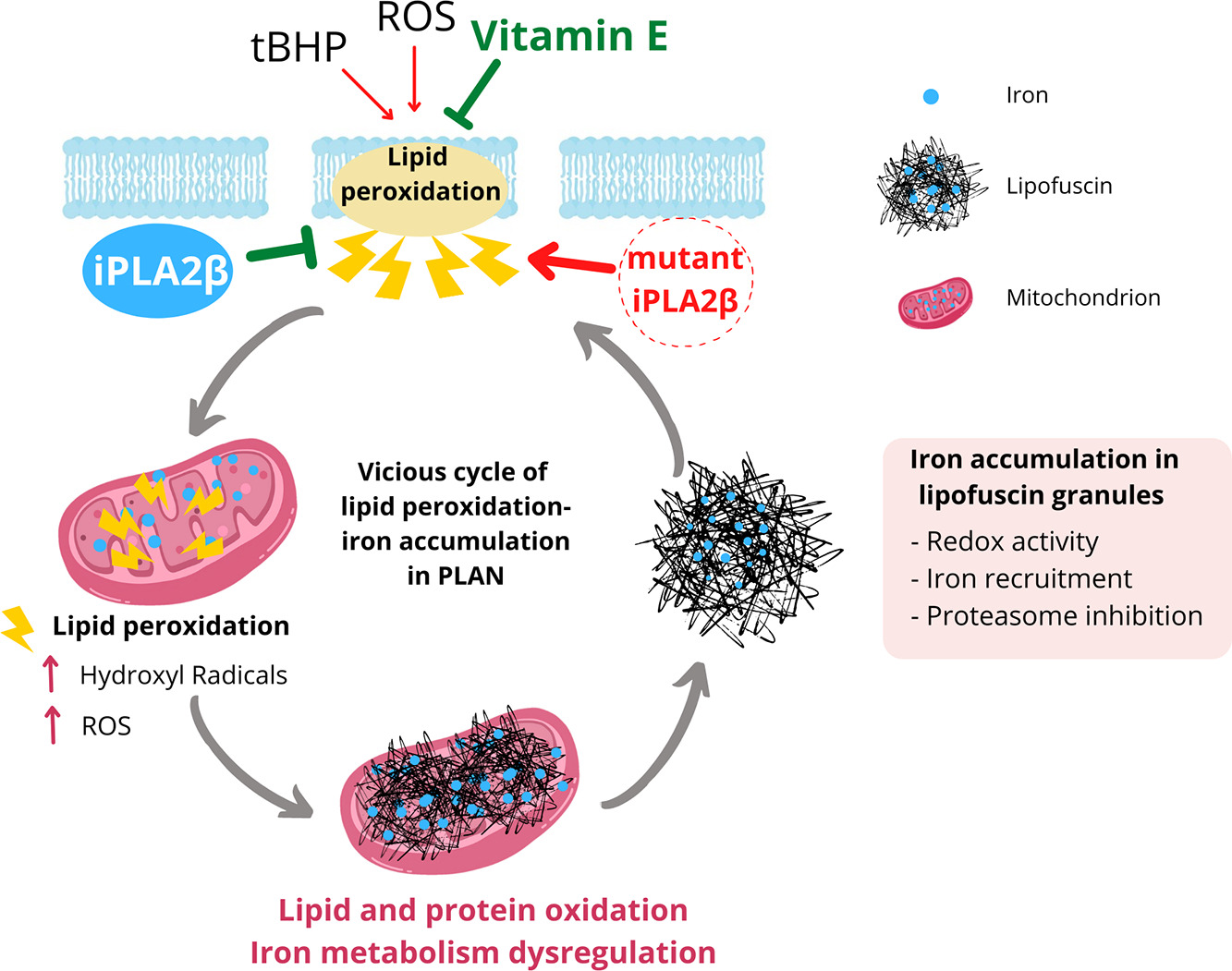
Lipid peroxidation as an inducer mechanism of iron accumulation
Friedreich’s Ataxia
Friedreich ataxia (FRDA) is an autosomal recessive disorder caused by deficiency of frataxin, an essential mitochondrial protein involved in iron sulfur cluster biogenesis, oxidative phosphorylation and other processes. FRDA most notably affects the heart, sensory neurons, spinal cord, cerebellum, and other brain regions, and manifests clinically as ataxia, sensory loss, dysarthria, spasticity, and hypertrophic cardiomyopathy.
Friedreich’s ataxia (FRDA) is caused by an intronic guanine-adenine-adenine (GAA) trinucleotide expansion in the gene encoding the frataxin protein (FXN). This triggers the transcriptional silencing of the frataxin gene (FXN) and subsequent FXN deficiency in affected cells, which accounts for the multisystemic symptoms of this condition. Current management strategies aim for symptomatic relief and no treatments can prevent disease onset or progression.
We are currently performing personalized medicine in more than 10 patients from both Spain and abroad.
Amyotrophic Lateral Sclerosis (ALS)
Amyotrophic Lateral Sclerosis is a neurodegenerative disease caused by progressive loss of motor neurons, which severely compromises skeletal muscle function. Evidence shows that muscle may act as a molecular powerhouse, whose final signals generate in patients a progressive loss of voluntary muscle function and weakness leading to paralysis. This pathology is the result of a complex cascade of events that involves a crosstalk among motor neurons, glia, and muscles, and evolves through the action of converging toxic mechanisms. In fact, mitochondrial dysfunction, which leads to oxidative stress, is one of the mechanisms causing cell death. There are two existing forms of the disease: sporadic and familial. Other factors include excitotoxicity, inflammation, and protein aggregation. Currently, the only approved drug for therapy is riluzole, that modestly prolongs survival. Thus, there is a need of effective treatments to reverse the damage in this devastating pathology. We are currently performing personalized medicine in 2 patients from which we hope to obtain really positive results.
Braincure for ARSACS (Autosomal recessive spastic ataxia of Charlevoix-Saguenay)
Autosomal recessive spastic ataxia of Charlevoix-Saguenay (ARSACS) is a slowly progressive condition with a variable clinical presentation. It is defined by a triad of early-onset cerebellar ataxia, spasticity, and sensorimotor polyneuropathy (characterized by distal muscle wasting and deformities in the fingers and feet). While the exact incidence of ARSACS remains uncertain, it has been estimated at 1 in 1,932 at birth. The gene associated with ARSACS, SACS, is located on chromosome 13q12 and encodes sacsin, a protein described in its canonical form as a polypeptide with 4,579 amino acids (GenBank accession no. NP_055178.3). Sacsin has been shown to participate in chaperone activities, regulate microtubule stability and cell migration, and likely play a key role in mitochondrial function regulation.
The primary objective of this project is to characterize the pathophysiological mechanisms in fibroblasts and neuronal cells from patients with SACS mutations. Additionally, it aims to evaluate the effectiveness of a library of commercially available drugs, including those commonly used to treat neurological conditions in clinical practice. The project is highly practical, focusing on identifying effective treatments for ARSACS patients.
Specific Aims of the BRAINCURE for ARSACS project:
- Characterization of the pathophysiology of ARSACS in fibroblasts derived from patients Selection of appropriate cellular alterations suitable for the evaluation of pharmacological screening rescues.
- High throughput screening of autophagy modulators and FDA approved drugs using fibroblasts derived from ARSACS patients. Confirmation of the beneficial effects of positive compounds on the pathophysiological alterations in mutant fibroblasts.
- Generation of induced neurons cells from control and ARSACS fibroblasts by direct reprogramming.
- Positive compounds in fibroblasts screening will be tested and evaluated on their ability to restore the pathological alterations in induced neuronal cells.
Braincure for Congenital Hypomyelinating Neuropathy-3 (CHN3)
Congenital Hypomyelinating Neuropathy-3 (CHN-3) is an autosomal recessive neurological disorder characterized by the onset of neurogenic muscle deterioration in utero. Affected individuals are born with severe hypotonia, often leading to respiratory failure and an inability to swallow or feed properly. They exhibit profound psychomotor impairment and may not survive beyond infancy or early childhood. Those who do survive are unable to sit or walk.
A sural nerve biopsy reveals hypomyelination of nerve fibers, while brain imaging frequently shows impaired myelination along with cerebral and cerebellar atrophy. Nerve conduction velocities are severely reduced (approximately 10 m/s) or even absent due to inadequate myelination.
Congenital Hypomyelinating Neuropathy-3 is associated with mutations in the CNTNAP1 gene (Contactin-associated protein 1), which plays a crucial role in myelination.
Specific Aims of the BRAINCURE for CHN3 project:
The main objectives of this project are:
1. Characterization of the pathophysiology of the disease in fibroblasts derived from CNTNAP1 patients.
2. Pharmacological screenings of UPR modulators, autophagy and mitochondrial booster.
3. Generation of induced neuronal cells from control fibroblasts and CNTNAP1 by direct reprogramming and confirmation of positive compounds found in fibroblasts screenings in one of the most affected cells in the disease.
Braincure for RAC1 mutations
RAC1 mutations cause ultra-rare and untreatable disorders that results in highly variable phenotypes and requires further investigation. RAC1 is part of the RAC Rho GTPases subfamily that also includes RAC2, RAC3, and RhoG; a class of molecules that modulate numerous cellular functions essential for normal development. RAC1 is an important modulator of the cytoskeleton, with a critical function in phagocytosis, mesenchymal-like migration, neuronal polarization, axonal growth, adhesion, and differentiation of multiple cell types. Additionally, it is involved in cellular growth and cell-cycle regulation via mTOR signaling. Individuals with RAC1 mutations result in varying degrees of developmental delay, brain malformations, and additional phenotypes, such as microcephaly, reduced neuronal proliferation, and cerebellar abnormalities in vivo.
Specific Aims of the RAC1 project:
The main objectives of this project are:
1. Characterization of the pathophysiology of the disease in fibroblasts derived from RAC1 patients.
2. Pharmacological screenings .
3. Generation of induced neuronal cells from control and RAC1 fibroblasts by direct reprogramming and confirmation of positive compounds found in fibroblasts screenings in one of the most affected cells in the disease.

Mitocure Project
The Mitocure project was developed for performing precision or personalized medicine in mitochondrial diseases.
Mitochondrial diseases are chronic (long-term), genetic, often inherited disorders that occur when mitochondria fail to produce enough energy for the body to function properly. Mitochondrial diseases can be present at birth, but can also occur at any age. Mitochondrial diseases can affect almost any part of the body, including the cells of the brain, nerves, muscles, kidneys, heart, liver, eyes, ears or pancreas, because mitochondria produce 90% of the energy our body needs to function.
Mitochondrial diseases include a group of chronic and progressive muscular and neurodegenerative disorders caused by a great variety of mutations in nuclear (nDNA) or mitochondrial DNA (mtDNA), most of which have no effective treatment. Current pharmacological therapies are based primarily on: (1) Eliminate toxic metabolites; (2) Try to circumvent the blockages of the respiratory chain; (3) Administer metabolites and cofactors to improve the synthesis of ATP; and (4) Prevent oxidative stress.
Given the diversity of mutations and the different therapeutic options, our proposal argues that a personalized therapeutic approach is required in mitochondrial diseases. In the Mitocure platform, we evaluate the therapeutic effectiveness of currently available treatments in fibroblasts derived from mitochondrial patients and in neuronal cells generated by direct reprogramming. To achieve this objective, we study the effects of these treatments on the pathophysiological alterations present in mutant cells. As a screening strategy, we examined cell proliferation and/or cell death in galactose culture medium (which forces energy to be obtained by the mitochondria), using an automated platform for live-cell imaging (Celldiscoverer 7, Zeiss). Combined analyses of phase-contrast and fluorescence images allow assessment of treatment effects on cell proliferation as well as the extent and kinetics of cell death. A pharmacology library of compounds frequently used in the treatment of mitochondrial patients is evaluated: antioxidants, AMP-activated protein kinase (AMPK) activators, autophagy/mitophagy modulators, mitochondrial dynamics modulators; inflammasome inhibitors; mitochondrial unfolded protein response (UPR) activators; and combinations of several treatments. Next, positive compounds are confirmed by assessing their effect on mitochondrial respiratory chain activity, expression levels of mitochondrial proteins, mitochondrial membrane potential, oxidative stress, and the activation of mitophagy and/or apoptosis in fibroblast and induced neurons iNs.
Currently, Mitocure platform is performing precision measurement in more than 20 mutations that directly affect mitochondrial oxidative phosphorylation. Some of the diseases included in the project are: Leigh syndrome, MELAS syndrome, Leber hereditary optic neuropathy (LHON), Mitochondrial complex I deficiency, mitochondrial type 3, Mitochondrial complex I deficiency, mitochondrial type 1, Mitochondrial complex I deficiency, nuclear type 5 (NDFS1), Mitochondrial complex IV deficiency, nuclear type 6 (COX15), Combined oxidative phosphorylation deficiency 1 (GFM1), Lipoyltransferase 1 deficiency (LIPT01), among others.
Mitocure-KAT6A Syndrome Platform
KAT6A syndrome is an extremely rare genetic neurodevelopmental disorder in which there is a variation (mutation) in the KAT6A gene. Variations in the KAT6A gene can potentially cause a wide variety of signs and symptoms; how the disorder affects one child can be very different from how it affects another. Neurodevelopmental disorders are ones that impair or alter the growth and development of the brain and the central nervous system. Common symptoms include varying degrees of intellectual disability, delays in reaching developmental milestones (developmental delays), delays in being able to speak (speech delays), and diminished muscle tone (hypotonia). Additional symptoms including abnormalities affecting the heart, eyes, and gastrointestinal system can also occur. In most instances, variations in the KAT6A gene occur spontaneously and there is no family history of the disorder (de novo variations). However, although researchers have been able to establish a clear syndrome with characteristic symptoms, much about KAT6A syndrome is not fully understood and there is not curative treatment, treatment is based on the specific symptoms present in each individual.
Recent hypotheses point out that mutations in the KAT6A gene affect secondary mitochondrial function and that drugs that act at the mitochondrial level (such as carnitine and vitamin B5) are capable of reversing the clinical phenotypes of the disease.
In the Mitocure-KAT6A platform, we evaluate the therapeutic effectiveness of several commercially available treatments that act at the mitochondrial level in fibroblasts derived from KAT6A patients and in neuronal cells generated by direct reprogramming. To achieve this objective, we study the effects of these treatments on the pathophysiological alterations present at the cellular level such as cell proliferation, mitochondrial respiratory chain enzymatic activities, coenzyme Q10 levels, mitochondrial protein expression levels, mitochondrial membrane potential, and activation of mitophagy and/or apoptosis.
We are currently performing personalized medicine in more than 20 patients from both Spain and abroad. This project receives funding from the KAT6A Association (Spain), the “KAT6A y Amigos” Association (Spain), the “Súper Auténticos” Association (Spain) and the KAT6A Foundation (US).
Mitocure-KAT6B Plantform
Soon, our research group will begin to carry out personalized medicine in cells from patients with KAT6B syndrome.
KAT6B is a rare genetic syndrome resulting from a mutation in the KAT6B gene. The KAT6B gene modifies structural proteins, which are important for the development of the nervous and skeletal systems. KAT6B disorders include genitopatellar syndrome (GPS) and Say-Barber-Biesecker-Young-Simpson variant of Ohdo syndrome (SBBYSS) which are part of a broad phenotypic spectrum with variable expressivity; individuals presenting with a phenotype intermediate between GPS and SBBYSS have been reported. Classically, these syndromes are distinct clinical entities caused by de novo (meaning that it was not inherited from either parent) truncation variants of the KAT6B gene. Of complete penetrance and variable expressivity, there are common phenotypic features and other characteristic ones. Both phenotypes are characterized by some degree of global developmental delay / intellectual disability; hypotonia; genital abnormalities; and skeletal abnormalities including patellar hypoplasia/agenesis, flexion contractures of the knees and/or hips, and anomalies of the digits, spine, and/or ribs. Congenital heart defects, small bowel malrotation, feeding difficulties, slow growth, cleft palate, hearing loss, and dental anomalies have been observed in individuals with either phenotype.
Mitocure-PACURE Platform – Propionic Acidemia
Propionic acidemia (PA) is a rare metabolic disorder that affects 1/20,000 to 1/250,000 people in various regions of the world. It is characterized by deficiency of propionyl-CoA carboxylase (PCC), an enzyme involved in the breakdown, catabolism of the amino acids valine, methionine, isoleucine, and threonine, and in the metabolism of cholesterol and odd-chain fatty acids. PCC deficiency leads to an elevation of propionyl-CoA, 3-hydroxypropionic acid, and ethylcitrate, ultimately leading to mitochondrial toxicity. Mutations in the PCCA or PCCB genes, which encode both subunits of propionyl-CoA carboxylase, cause propionic acidemia with autosomal recessive inheritance. Symptoms occur most often during the first weeks of life and may include abnormally decreased muscle tone (hypotonia), poor feeding, vomiting, listlessness (lethargy), dehydration, and seizures. People with this condition must follow a specific diet that includes a low protein intake and specific food formulas.
Specific objectives of the PACURE project:
1) Characterization of the pathophysiology of fibroblasts derived from patients with PA.
2) Drug screening in fibroblasts derived from PA patients. Evaluation of drugs capable of improving pathophysiological alterations. Likewise, we will study the underlying molecular mechanisms in the action of drugs with positive effects.
3) Generation of neuronal cells by direct reprogramming of fibroblasts from PA patients.
4) Positive compounds in the fibroblast screening will be evaluated in the differentiated neuronal cells.
Mitocure-EECURE Platform – Ethylmalonic Encephalopathy
This research project consists of carrying out personalized medicine for the Ethylmalonic Encephalopathy disease. Specifically, this project begins with the financing of the association “Todos con Adán”, the only patient in Spain diagnosed with this rare disease.
Ethylmalonic encephalopathy (EE) is an extremely rare disease caused by a recessive defect of the ETHE1 gene; it presents in infancy and follows a progressive course characterised by psychomotor retardation, hypotonia, and generalised microvascular damage. Specifically, EE is a rare metabolic and mitochondrial disorder caused by dysfunction of ETHE1 protein, a mitochondrial dioxygenase involved in hydrogen sulfide (H2S) detoxification. High levels of ethylmalonic acid are detected in the body fluids, and cytochrome c oxidase activity is decreased in skeletal muscle. Delayed growth with diarrhoea and dysphagia is a common symptom. Also neurological impairment is accelerated in the context of intercurrent infectious diseases. Children may be unable to walk without support and may be wheelchair bound and patients typically do not live beyond the first decade of life. Nowadays treatment includes antioxidants, antibiotics that lower H2S levels and antispastic medications, which are not curative.
So, our main objective is to find alternative treatments for patients with this disease.

CancerCure Project
The main objective of this project is to look for personalized treatments for genetic susceptibility for cancer.
The term “cancer susceptibility” describes the higher risk of cancer that certain people have compared to the risk for the general population. People may have a very high risk linked to a strong family history (e.g., 50–90% chance of acquiring cancer) or a considerably elevated relative risk (e.g., 2–10 times the population risk), which is nonetheless associated with a very low overall risk for cancer. A substantial hereditary component plays a role in the illness in both situations.
Li-Fraumeni Syndrome (LFS)
Li-Fraumeni syndrome (LFS) is a rare, inherited, cancer predisposition syndrome characterized by the early start of several primary malignancies, such as brain tumors, leukemias, soft tissue and bone sarcomas, breast cancer, and adrenal cortical carcinoma (ACC). Individuals with LFS are around 50% more likely to have another malignancy. It is unclear how often pathogenic and potentially pathogenic germline TP53 mutations are, however, estimates range from 1/3,555 to 5,476. Mutations in the tumor suppressor gene TP53 (17p13.1) are the etiology of LFS in most families with typical LFS. The p53 protein, which is involved in several biological functions such DNA repair, growth arrest, and apoptosis, is encoded by this gene. De novo TP53 mutations are thought to account for 7–20% of cases.
Ataxia-telangiectasia (A-T)
Ataxia-telangiectasia (A-T) is a is an autosomal recessive disorder that causes loss of muscle control and balance, cancer, lung disease and immune system problems in children and young adults. It has been estimated that 1 in every 40.000-100.000 babies are born with A-T. It is characterized mainly by cerebellar degeneration, telangiectasia, immunodeficiency, cancer susceptibility and radiation sensitivity. The overall objective of this project is to characterize the pathophysiological mechanisms in fibroblasts and neuronal cells derived from patients with ATM (Ataxia Telangiectasia Mutated) genetic mutations and to rescue the pathophysiological alterations using a library of FDA-approved drugs.
